Accelerating clinical development for emerging biotech sponsors
Faster startup • Direct execution • Efficient development
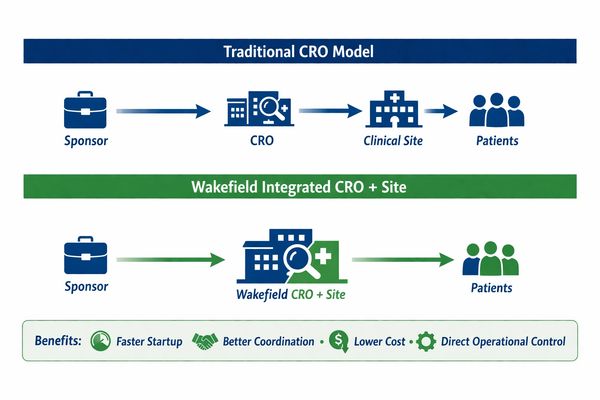
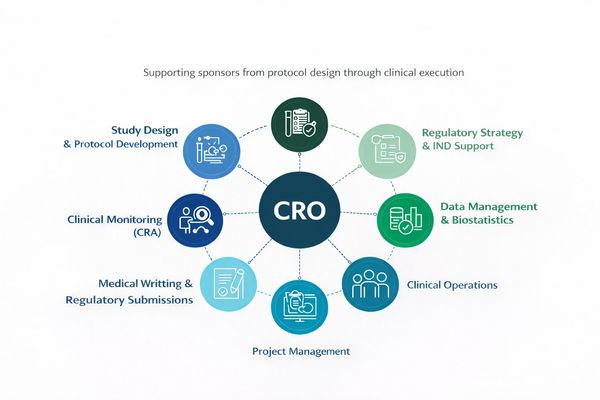
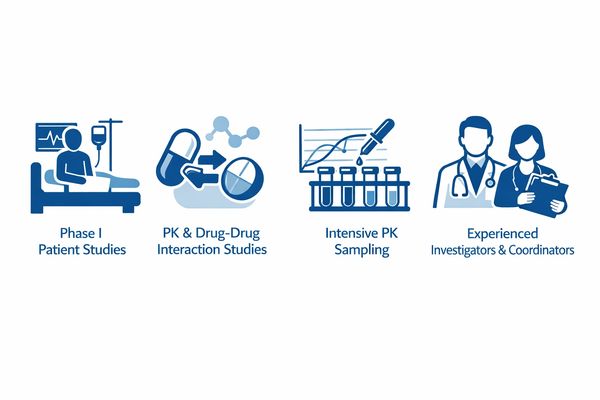
Integrated CRO + Clinical Research Site
Accelerating clinical development for emerging biotech sponsors
Faster startup • Direct execution • Efficient development